Acetal-Linked Paclitaxel Prodrug Micellar Nanoparticles as a Versatile and Potent Platform for Cancer Therapy
Yudan Gu , Yinan Zhong , Fenghua Meng *(孟凤华), Ru Cheng , Chao Deng , and Zhiyuan Zhong * (钟志远)
Biomedical Polymers Laboratory and Jiangsu Key Laboratory of Advanced Functional Polymer Design and Application, Department of Polymer Science and Engineering, College of Chemistry, Chemical Engineering and Materials Science, Soochow University, Suzhou 215123, P. R. China
Biomacromolecules, 2013, 14, 2772–2780.
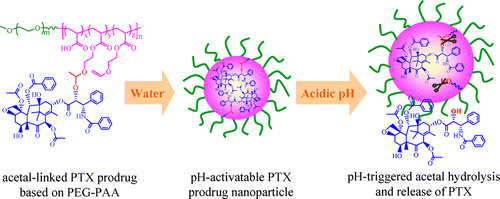
Endosomal pH-activatable paclitaxel (PTX) prodrug micellar nanoparticles were designed and prepared by conjugating PTX onto water-soluble poly(ethylene glycol)-b-poly(acrylic acid) (PEG-PAA) block copolymers via an acid-labile acetal bond to the PAA block and investigated for potent growth inhibition of human cancer cells in vitro. PTX was readily conjugated to PEG-PAA with high drug contents of 21.6, 27.0, and 42.8 wt % (denoted as PTX prodrugs 1, 2, and 3, respectively) using ethyl glycol vinyl ether (EGVE) as a linker. The resulting PTX conjugates had defined molecular weights and self-assembled in phosphate buffer (PB, pH 7.4, 10 mM) into monodisperse micellar nanoparticles with average sizes of 158.3–180.3 nm depending on PTX contents. The in vitro release studies showed that drug release from PTX prodrug nanoparticles was highly pH-dependent, in which ca. 86.9%, 66.4% and 29.0% of PTX was released from PTX prodrug 3 at 37 °C in 48 h at pH 5.0, 6.0, and pH 7.4, respectively. MTT assays showed that these pH-sensitive PTX prodrug nanoparticles exhibited high antitumor effect to KB and HeLa cells (IC50 = 0.18 and 0.9 μg PTX equiv/mL, respectively) as well as PTX-resistant A549 cells. Notably, folate-decorated PTX prodrug micellar nanoparticles based on PTX prodrug 3 and 20 wt % folate-poly(ethylene glycol)-b-poly(d,l-lactide) (FA-PEG-PLA) displayed apparent targetability to folate receptor-overexpressing KB cells with IC50 over 12 times lower than nontargeting PTX prodrug 3 under otherwise the same conditions. Furthermore, PTX prodrug nanoparticles could also load doxorubicin (DOX) to simultaneously release PTX and DOX under mildly acidic pH. These acetal-linked PTX prodrug micellar nanoparticles have appeared as a highly versatile and potent platform for cancer therapy.
链接: //pubs.acs.org/doi/abs/10.1021/bm400615n